Our Relationship with HIF-1α; its Complicated
766 Words | 3 Min Read
The human body has an intricate relationship with the naturally occurring protein HIF-1α, which is vital for life, but imbalances in its expression can result in life-threatening conditions. HIF-1α, or hypoxia-inducible factor-1 alpha, plays a critical role in the cellular response to low oxygen levels. HIF-1α is crucial in helping our body adapt to low oxygen levels, known as hypoxia. HIF-1α is a transcription factor that responds to changes in oxygen availability in the cellular environment.
Under hypoxic conditions, HIF-1α stabilizes and forms a complex with HIF-1β. This complex then binds to hypoxia-response elements in the DNA, activating various genes involved in several adaptive processes. These processes include angiogenesis (formation of new blood vessels), erythropoiesis (production of red blood cells), glycolysis (anaerobic metabolism of glucose), and other mechanisms that help cells and tissues maintain oxygen homeostasis.
In my world, where N1 is worried about inflammatory lung diseases such as acute respiratory distress syndrome (ARDS), HIF-1α can have both beneficial and detrimental effects, making it difficult to classify it solely as a “friend” or “foe.”
On the one hand, HIF-1α can help the body adapt to the reduced oxygen levels often associated with pneumonia or other lung conditions. HIF-1α can help improve oxygen delivery and utilization in affected tissues by promoting processes like angiogenesis, erythropoiesis, and glycolysis.
(Image credit: xia yuan via Getty Images)
On the other hand, evidence suggests that HIF-1α can play a role in the inflammatory response in pneumonia and other lung diseases. Inflammation is a necessary part of the body’s immune response to infection, but excessive or prolonged inflammation can damage lung tissue and worsen the disease. This is the case sometimes in ARDS. Some studies have shown that HIF-1α can contribute to producing pro-inflammatory cytokines and recruiting immune cells to the site of infection, which could exacerbate lung inflammation. This very much describes the terror we all saw in the early days of the COVID-19 pandemic.
Overexpression of HIF-1α is also associated with various diseases, including cancer, cardiovascular disorders, and inflammation. Although some promising therapies exist, many current pharmaceutical approaches rely on inhibiting rather than regulating and repurposing existing medications.
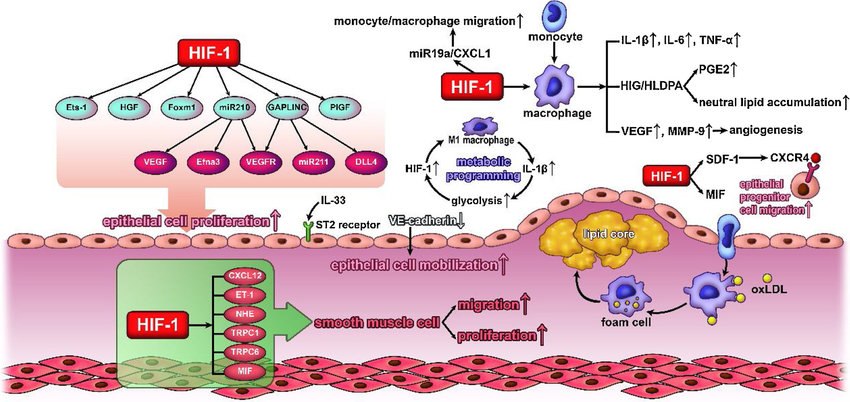
Image credit: Biomedicine & Pharmacotherapy Oxygen homostasis and cardiovascular disease: A role for HIF? August 2020
Examples of HIF-1α-targeting treatments include inhibitors like PX-478, YC-1, and chetomin, and upstream regulators such as PI3K/Akt/mTOR pathway inhibitors rapamycin and everolimus. Alternatively, some therapies adopt a downstream approach, targeting proteins regulated by HIF-1α, such as bevacizumab and sunitinib, to inhibit new blood vessel formation.
Innovative approaches involve using antisense oligonucleotide therapy, which employs synthetic nucleic acid sequences to bind mRNA molecules and inhibit protein translation or utilizing RNA interference (RNAi) to induce protein degradation. Another versatile (and, in the author’s mind’s eye, overhyped) strategy includes gene editing, specifically CRISPR/Cas9, in this context.
The fact is that HIF-1α is a naturally occurring protein with a crucial function, and its under-expression can lead to its own troubles, including impaired erythropoiesis, causing anemia, insufficient angiogenesis that affects tissue regeneration and wound healing, and weakened immune responses, impacting macrophages, neutrophils, and the production of inflammatory cytokines.
HIF-1α is essential in our body’s natural defense against low oxygen conditions. Often, maintaining a balance seems more fitting than an aggressive approach. Consistent with N1’s philosophy, we strive for equilibrium, overall homeostasis, and a return to normalcy.
As we progress in our preclinical development of a therapy for ARDS, we’ve observed indications that our proprietary NV1 protein provides additional rescue from hypoxic conditions in lung tissue, heightening the already numerous regenerative effects of the native NELL-1 protein inherent in NV1, such as balancing cytokines, restoring damaged tissue and encouraging blood vessel formation.
Our research on NV1’s rescue from hypoxic conditions has yielded promising results as we strive to develop comprehensive pharmaceutical solutions that guide the body back to homeostasis.
In our pursuit of an ARDS therapeutic, human bronchial epithelial cells were subjected to hypoxic conditions and treated with native NELL-1 and NV1 while an appropriate control was utilized. Following cell lysis, the protein levels of HIF-1α were quantified with β-Actin as a housekeeping loading control. The study found that NV1 was significantly more effective than both native NELL-1 and untreated cells in modulating HIF-1α levels from highly elevated levels to near-normal levels. These favorable results demonstrate the potential of NV1 as a powerful pharmaceutical intervention for ARDS patients and highlight its superiority over native NELL-1 as a therapeutic.
Numerous remarkable ongoing research and development efforts have the potential to revolutionize medicine and save lives, marking an extraordinary era in human history for drug development. N1 is grateful to be part of this vibrant scientific community and remains committed to pushing the boundaries of science while prioritizing the well-being of humanity over profit.


